Climate change is one of the most pressing issues of our time.
Since the mid-1800s, humans have played a major role in releasing greenhouse gases into the atmosphere, primarily through the burning of oil, gas, and coal for heating homes, powering factories, and transportation.
Greenhouse gases, primarily carbon dioxide (CO2), trap the sun’s heat and cause the planet’s temperature to rise. Since the nineteenth century the amount of CO2 in the atmosphere has increased by 50% and the world is approximately 2F warmer than it was in the late 1800s.
In the United States, passenger cars and light trucks produce approximately 20% of total greenhouse gas emissions. Switching from a gas to an electric vehicle is one of the single best ways you can reduce your carbon footprint and help make an immediate positive impact on the environment.
 Electric Vehicles are better for the environment because they produce significantly fewer emissions than gas-powered cars. This is true throughout their entire lifecycle, not just while in use. This makes EVs powerful tools in the fight against climate change, which is more important than ever. With no tailpipe emissions, EVs also reduce air pollution, which is especially important for the most vulnerable communities who are disproportionately harmed by air pollutants.
Electric Vehicles are better for the environment because they produce significantly fewer emissions than gas-powered cars. This is true throughout their entire lifecycle, not just while in use. This makes EVs powerful tools in the fight against climate change, which is more important than ever. With no tailpipe emissions, EVs also reduce air pollution, which is especially important for the most vulnerable communities who are disproportionately harmed by air pollutants.
According to the US Environmental Protection Agency, 20 pounds of CO2 is produced for every gallon of gasoline burned in an internal combustion engine. A relatively efficient internal combustion engine car that gets 25 miles per gallon and drives 12,000 miles per year will produce 9,600 pounds of CO2 during that time. Even a very efficient plug in hybrid that gets 42 miles per gallon will still produce 5,700 pounds CO2 while driving 12,000 miles in a year. Replacing even 10% of gas powered cars in the U.S. with electric cars would remove 173 billion pounds of CO2 from the atmosphere every year, which will go a long way towards repairing the damage from climate change.
VEOS’ car sharing subscription model results in higher utilization for each car, resulting in fewer overall cars on the road and even greater greenhouse gas reductions.
Additionally, our highly efficient direct to consumer online subscription model has a lower carbon footprint than the traditional franchise dealer sales and distribution model, resulting in further greenhouse gas reductions.
As a financial consultant, I travel often to see clients. My prized possession is a classic 1955 MG TD. I decided to sign up for the VEOS subscription plan for 20 days a month to keep my restored classic car in pristine shape. It’s cheaper than traditional renting and leasing by far.

How can one gallon of gasoline produce twenty pounds of CO2?
It seems impossible that a gallon of gasoline, which weighs about 6.3 pounds, could produce 20 pounds of carbon dioxide (CO2) when burned. However, most of the weight of the CO2 doesn’t come from the gasoline itself, but the oxygen in the air.
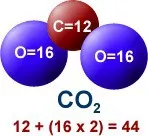 When gasoline burns, the carbon and hydrogen separate. The hydrogen combines with oxygen to form water (H2O), and carbon combines with oxygen to form carbon dioxide (CO2).
When gasoline burns, the carbon and hydrogen separate. The hydrogen combines with oxygen to form water (H2O), and carbon combines with oxygen to form carbon dioxide (CO2).
A carbon atom has a weight of 12, and each atom has a weight of 16, giving each single molecule of CO2 an atomic weight of 44 (12 from carbon and 32 from oxygen).
Therefore, to calculate the amount of CO2 produced from a gallon of gasoline, the weight of the carbon in the gasoline is multiplied by 44/12 or 3.7.
Since gasoline is about 87% carbon and 13% hydrogen by weight, the carbon in a gallon of gasoline weighs 5.5 pounds (6.3 lbs. x .87).
We can then multiply the weight of the carbon (5.5 pounds) by 3.7, which equals 20 pounds of CO2!
Data Sources: Physical and chemical properties of gasoline: Department of Energy (DOE), Alternative Fuels Data Center (AFDC), Properties of Fuels.